- Blog
- Hanoi rocks buried alive
- The missing link pet kelp formula skin and coat
- Beserk in hyper bullet
- Solarwinds breach
- Mmass of hexane
- Sugar rush addison grace lyrics
- Race driver grid nds rom
- Mind control porn comics
- You need a budget message board
- Qview qd700 cheat sheet
- Utorrent alternative
- 2021 moscow victory day parade
- Blog
- Hanoi rocks buried alive
- The missing link pet kelp formula skin and coat
- Beserk in hyper bullet
- Solarwinds breach
- Mmass of hexane
- Sugar rush addison grace lyrics
- Race driver grid nds rom
- Mind control porn comics
- You need a budget message board
- Qview qd700 cheat sheet
- Utorrent alternative
- 2021 moscow victory day parade
You can see how this formula works reading the example above or/and choosing another one at the bottom of this page. Chloroform, n-hexane, and n-hexane-ethyl acetate showed the. That is exactly the formula that we use in this calculator.
MMASS OF HEXANE HOW TO
M kg v m³ = m lb v gal × mcf vcf, but m kg v m³ is, by definition, the density (d) in kg/m³, soįinally, we can generalize this formula any units of mass and volume, since we know how to find the values for mcf and vcf (Check the factor tables below). Now, dividing the equation (1) by the equation (2), we get
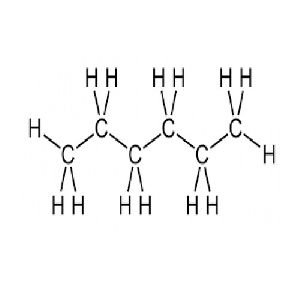
Volume in cubic meters = volume in gallons × vcf or We will call 0.003785411784 the volume conversion factor - vcf To transform the volume from gallons to cubic meters we should multiply the volume in cubic meters by 0.003785411784. Answer to Solved Mass of hexane density volume Science Chemistry Chemistry questions and answers Mass of hexane density volume 0.660 g/mL 1000 mL 660 g Moles of hexane 660 g / 86.18 g/mol 7.66 moles 2 C6H14 + 19 O2 -> 14 H2O + 12 CO2 So moles of oxygen necessary 7. Mass in kilograms = mass in pounds × mcf or We will call 0.45359237 the mass conversion factor - mcf To transform the mass from pounds to kilograms we should multiply the mass in pounds by 0.45359237. If we have, for example, mass in pounds and volume in gallons, we should do something to correct our result. For example, mass in kg and volume in liters. But this works fine when the density value we have (from a chart for example) is defined using the same unit of volume and mass that we will use in the formula. 1 7 8 m o l g To find the molar mass of Hexane, we can look up the atomic masses from the Periodic table for each element that make up Hexane and multiply each by the number of atoms in the formula and then adds up everything. The DLOP is measured as mass per sample and expressed as equivalent air. Very simple isn't it? Yes, it is as simple as that. Hexane, whose chemical formula is C 6 H 1 4 has a molar mass of 8 6. Hexane is three times as acutely toxic to mice as is pentane concentrations of. Mass = density × volume = 750 × 2 = 1500 Kg Plugging the values in the formula, we get Well, by definition of density we just need to use the formula:Įxample: How much does 2 cubic meters of gasoline weigh in kilograms if the gasoline density is 750 kg/m³? How to convert from volume to mass (incorrectly called weight)?
Volume to Mass Converter (Construction).

